Mapping the Alzheimer’s proteome – Applications of proteomics in neuroscience

Tyler Ford
November 14, 2023
Alzheimer’s disease is a complex neurodegenerative disorder involving many brain regions, many stages of disease, and many pathological features (e.g. amyloid plaques, neurofibrillary tangles [NFTs], and cerebral amyloid angiopathy [CAA]). This complexity has been captured in the hundreds of proteomic studies of Alzheimer’s and the even more numerous publications on the roles of individual proteins in the disease. However, each individual study provides a narrow perspective on Alzheimer’s disease as most are limited by small sample sizes, the inclusion of few brain regions, or the inclusion of few Alzheimer’s disease stages.
A comprehensive study encompassing all stages of Alzheimer’s disease and all brain regions sounds like a monumental task especially if you take into account the number of samples you’d need for such an experiment. Rather than start from scratch, a recent study took another approach: a systematic literature review of multiple proteomics studies.
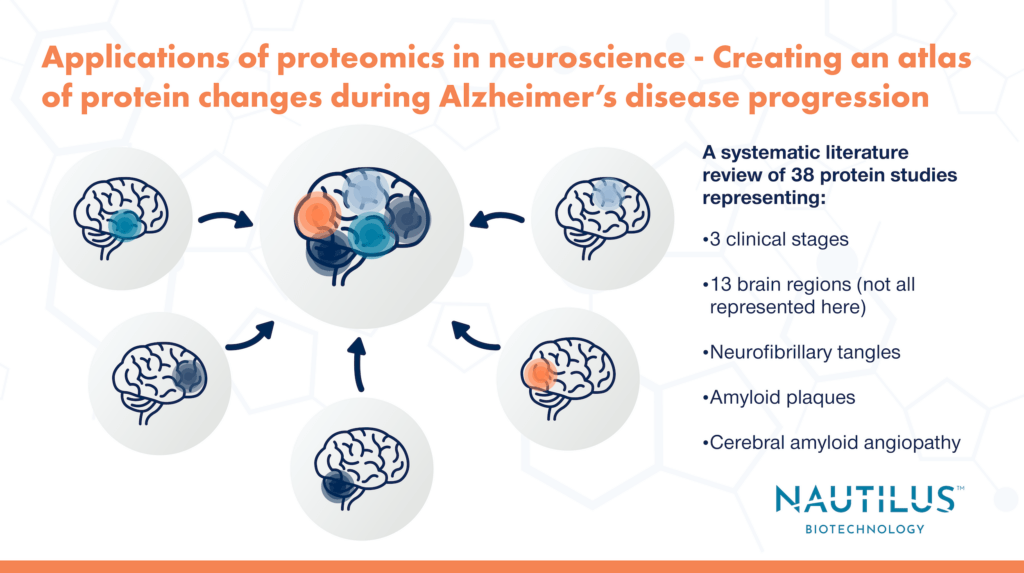
Askenazi et al. selected 38 publications using proteomics to study Alzheimer’s disease in humans. With data from these publications in hand, they set out to determine how the proteome changes across 3 clinical stages of disease, 13 brain regions, and 3 pathological features (or “lesions”). The amalgamation of data from their systematic review (which can be viewed in the NeuroPro database) allowed them to answer questions that would have been difficult with just one study on its own, for example:
- What proteins are enriched in amyloid plaques, NFTs, and cerebral amyloid angiopathy?
- What are the earliest protein changes in Alzheimer’s disease?
- How do protein abundance changes associated with Alzheimer’s disease vary between brain regions during Alzheimer’s progression?
Below, we take a dive into the data to see how the authors’ systematic review answers these questions and discuss how next-generation proteomics platforms like the NautilusTM Proteome Analysis Platform can propel future studies of Alzheimer’s and other diseases.
Check out our round up of proteomics and neuroscience posts.
Watch this animation to learn how next-generation proteomics technologies can fuel neuroscience
Common proteome alterations in Alzheimer’s disease identified across multiple studies
In their review, Askenazi et al. found 54 proteins were differentially expressed in at least 15 of the studies included. Because they were altered in so many studies encompassing different techniques, brain regions, and clinical stages of disease, it’s likely these are the most prevalent proteome changes in Alzheimer’s disease. These differentially expressed proteins provide a great opportunity for future scientific inquiry: 46% of these proteins are currently understudied in Alzheimer’s (≤ 10 previous publications) and 4 of these proteins have never been examined in the context of Alzheimer’s.
Proteins Enriched in Amyloid Plaques, NFTs, and CAA in Alzheimer’s Disease
While the 54 differentially expressed proteins mentioned above highlight widespread changes in Alzheimer’s disease, focusing on specific, localized features of the Alzheimer’s brain can help identify proteins involved in their formation. Here, the authors identified many protein expression changes associated with amyloid plaques, NFTs, and CAA. They found that only a few proteins were enriched in all three while the majority of the proteins enriched/depleted in each pathological feature likely reflected the processes involved in that feature’s formation.
These findings demonstrate the importance of doing proteomic studies on localized brain features in addition to bulk tissue samples. In bulk tissue experiments, the enrichment of these proteins could have been missed.
Temporal progression of protein dysfunction across brain regions in Alzheimer’s Disease
With their systematic review, Askenazi et al. could simultaneously examine proteome changes during different stages of disease. They could also examine different regions of the brain known to be “vulnerable” or “resistant” to pathological changes that occur during Alzheimer’s disease. For example, A𝛃 and tau (proteins that aggregate to form amyloid plaques and NFTs, respectively) are known to progressively spread throughout the brain affecting vulnerable regions first.
One of the key findings from their analysis was there is a widespread decrease in synapse-related protein expression in early Alzheimer’s suggesting general synaptic dysfunction. These changes occur before A𝛃 and tau accumulation and are likely the initiating drivers of disease. They may also be potential drug targets for therapeutics treating early Alzheimer’s.
Overall, Askenazi et al. found 258 altered proteins in early and late Alzheimer’s with very few proteins that trended in opposite directions in early vs. advanced stages (i.e. proteins that were highly expressed early generally remained highly expressed and vice versa). Furthermore, they saw the same patterns of protein changes in resistant brain regions in advanced disease as vulnerable brain regions in early disease. This patterning hints at a “temporal wave of progressive protein dysfunction” throughout the brain where resistant brain regions undergo the same changes as vulnerable regions, just at a later time.
Advancing Alzheimer’s disease research with next-generation proteomics platforms
Askenazi et al. uncovered several key proteomic changes in the progression of Alzheimer’s disease in different brain regions, in different neuropathological features, and at different stages in disease progression. As the authors demonstrated, examining the collection of proteomics publications for a given disease or biological process can paint a comprehensive picture of the entire proteomic landscape that would not have been possible with just one proteomic study alone.
However, the authors did note that some of the past proteomic studies included in the analysis were “underpowered.” With low-throughput, underpowered studies, low abundance proteins could be missed and there could be higher variability in the data. Rapid throughput, next generation platforms, such as the Nautilus Proteome Analysis Platform, can drive the detection of rare proteome changes, capture more data, and bring even more power to systematic literature reviews of protein changes in Alzheimer’s and beyond.
MORE ARTICLES
